OVERVIEW
Aquaculture, including shrimp farming, is the fastest-growing animal food production sector globally. Several emerging diseases – including Acute Hepatopancreatic Necrosis Disease (AHPND), Viral Covert Mortality Disease (VCMD), Enterocytozoon hepatopenaei (EHP) infection, and SHIV disease – have impacted the global shrimp farming industry. Recently, a new disease known as “post-larval body opacity” (TPD) or “glassy post-larval disease” (GPD) (hereinafter referred to as Transparent Post-larval Disease - TPD) in Pacific white shrimp ( Litopenaeus vannamei ) has become a rapidly increasing threat to shrimp farming in China.
Since March 2020, numerous cases of Transparent Post-larval Disease have occurred in Pacific white shrimp in hatcheries in Guangdong and Guangxi provinces. Subsequently, this new disease began to spread to major shrimp farming regions in northern China through the transportation of shrimp post-larvae in April 2020. Transparent Post-larval Disease primarily affects shrimp post-larvae aged 4 to 7 days old (PL4 to PL7) with a very high infectivity. Typically, the morbidity rate of an affected population can reach up to 60% on the second day after the first observation of abnormal individuals, and even up to 90-100% in severe cases by the third day. Shrimp affected by Transparent Post-larval Disease mainly exhibit similar clinical signs, such as pale or colorless hepatopancreas and an empty digestive tract, which makes the bodies of affected individuals transparent and opaque.
Transparent Post-larval Disease has become prevalent in farmed shrimp populations, leading to severe economic losses in some shrimp farming regions in China, thus making investigation and the development of disease prevention strategies urgent. The research team sampled TPD-affected shrimp and screened for several known shrimp pathogens, but the results indicated that these shrimp were free of known viral pathogens and suggested that Transparent Post-larval Disease might be caused by a newly emerging pathogen. Additionally, some farmers observed that treating pond water with antibacterial agents could reduce the disease, indicating that Transparent Post-larval Disease might be caused by a bacterial pathogen.
STUDY SETUP
Diseased L. vannamei shrimp (PL7, body length 6 to 8 mm) were sampled from a shrimp farm in Ganyu, Jiangsu Province, China. These were processed for histopathological examination and for observation by transmission electron microscopy. Healthy PL3 post-larvae (body length 4 to 6 mm) were purchased from a shrimp farm in Weifang, Shandong Province, and cultured for two days, then used for the experiment.
RESULTS AND DISCUSSION
AHPND occurs approximately 35 days after stocking shrimp into ponds, and the onset of clinical signs and mortality rates for AHPND begin as early as 10 days post-stocking in some severe cases. However, TPD typically occurs in PL4-7, much earlier than when AHPND usually appears. Therefore, we infer that TPD is a new disease distinct from AHPND.
Results from molecular tests showed that diseased individuals in post-larval rearing tanks affected by TPD were negative for major farmed shrimp diseases, including White Spot Syndrome Virus (WSSV), Infectious Hypodermal and Hematopoietic Necrosis Virus (IHHNV), AHPND, Enterocytozoon hepatopenaei (EHP), SHIV disease virus, Yellow Head Virus (YHV), Taura Syndrome Virus (TSV), and Infectious Myonecrosis Virus (IMNV).
Therefore, considering that treating water in post-larval rearing tanks with antibacterial agents could reduce the disease, we first proceeded to isolate and identify bacterial pathogens from samples of L. vannamei affected by Transparent Post-larval Disease. The predominant bacterium isolated from severely infected shrimp was identified as V. parahaemolyticus through various tests.
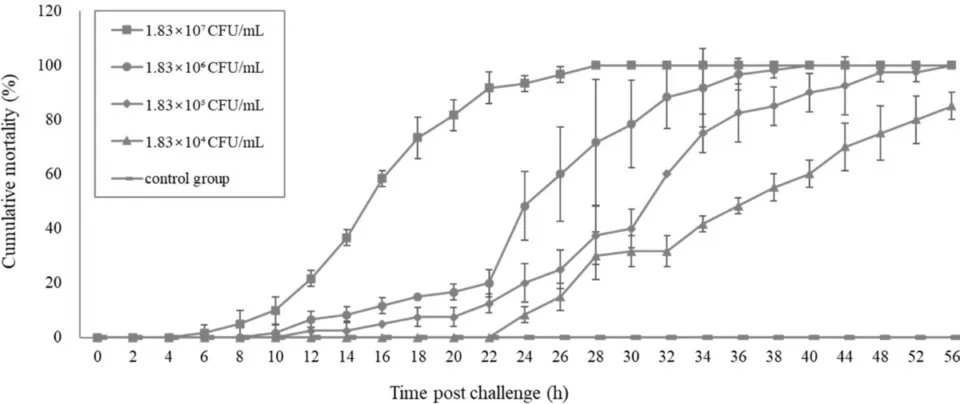
And the analysis of the pathogenicity of these V. parahaemolyticus in an experimental treatment indicated that the overall typical clinical signs were similar to those in farmed shrimp with Transparent Post-larval Disease. The onset and progression of the disease in the treatment, as well as the mortality rate, were also similar to those in farmed shrimp with Transparent Post-larval Disease. Therefore, this new V. parahaemolyticus species (Vp-JS20200428004-2) was inferred to be the causative agent of Transparent Post-larval Disease in shrimp post-larvae.
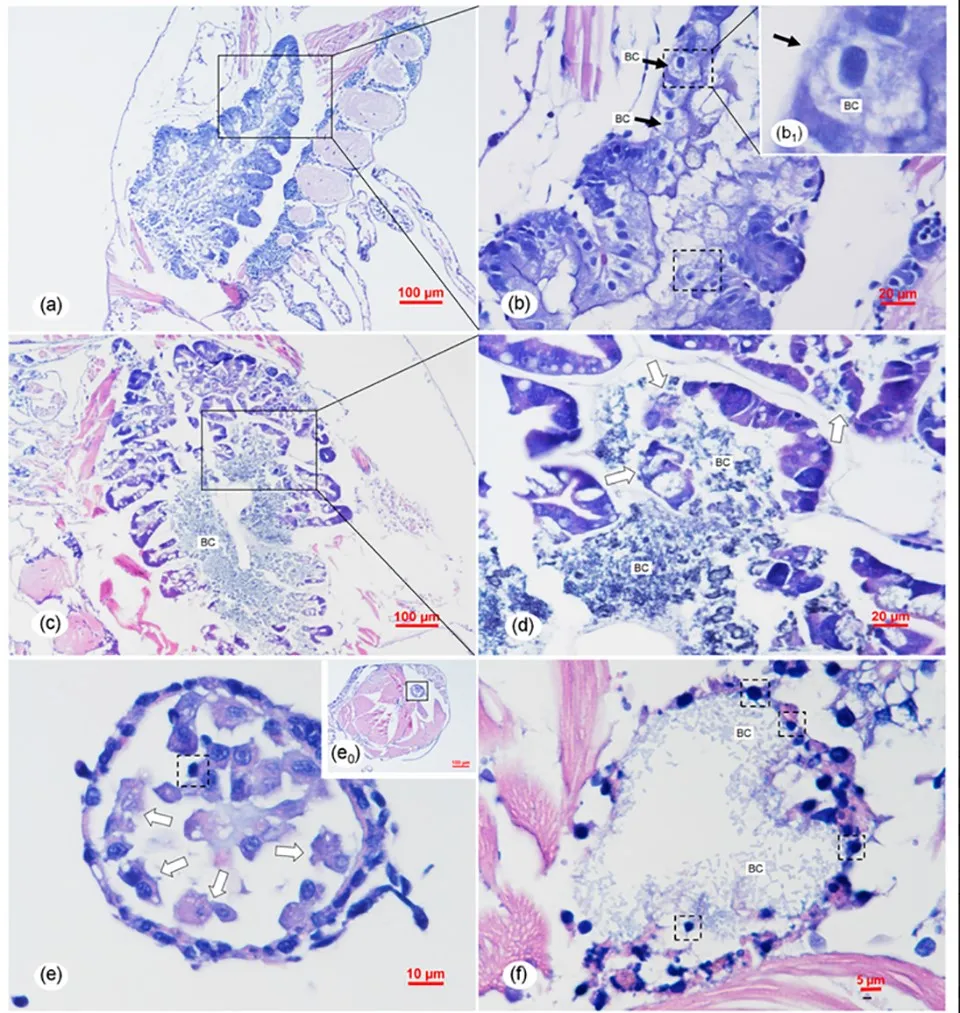
Based on the study results, the overall histopathological signs of Transparent Post-larval Disease are not similar to those of AHPND and also differ from those caused by atypical V. parahaemolyticus. To further explore the pathogenic mechanism of Vp-JS20200428004-2, more systematic comparative studies should be designed and conducted; detailed information on the functional genes, plasmids, and genome of Vp-JS20200428004-2 needs further investigation in the future.
To date, Vibriosis, caused by various Vibrio spp., has been reported to occur at different developmental stages of L. vannamei, including nauplius (2 days), zoea (4 to 5 days), mysis (3 to 5 days), post-larvae (10 to 15 days), and juveniles. Vibrio alginolyticus has been associated with zoea 2 syndrome and mysis mold syndrome at the larval stage, while V. alginolyticus and V. harveyi have been linked to bolitas syndrome.
The latter refers to a larval syndrome in L. vannamei involving the detachment of epithelial cells from the intestine and hepatopancreas, which appear as small spheres in the digestive tract. Clearly, the histopathological syndrome of the intestine and hepatopancreas in Transparent Post-larval Disease caused by Vp-JS20200428004-2 differs from bolitas syndrome. Additionally, the clinical signs of Transparent Post-larval Disease differ from those of bolitas syndrome, which include reduced feeding, stunted growth, slow swimming, and reduced excretion. Therefore, based on the study results, Transparent Post-larval Disease can be identified as a new Vibrio disease occurring at the PL stage of L. vannamei.
PERSPECTIVE
Based on the results of a systematic analysis including isolation, identification, and pathogenicity testing according to four criteria designed to establish the pathogenic relationship between bacteria and disease, it can be confirmed that a new V. parahaemolyticus species (Vp-JS20200428004-2) is the causative agent associated with the emerging Transparent Post-larval Disease affecting shrimp farming in China in 2020.
The new pathogen exhibits high virulence to shrimp post-larvae and can cause acute and severe histopathological changes in the hepatopancreas and digestive tract. The risk of epidemics and PL losses caused by this new pathogen warrants further attention.
This article is an abridged translation from the original publication by Zou, Y. et al., 2020 - Identification of the Infectious Agent of Transparent Post-larval Disease (TPD) in Penaeus vannamei






