This review details recent findings related to EHP infection in shrimp farms, including its effects on shrimp immunity, digestion, metabolism, physiology, and growth.

Dr. Allan Heres pathologist and shrimp health management specialist, Phibro Aqua

Shrimp infected with EHP
Hepatopancreatic microsporidiosis was first discovered in 2009 and is now prevalent in many major shrimp farming countries, where it has affected economic sustainability, production, profitability, and global shrimp supply. The disease is caused by Enterocytozoon hepatopenaei (EHP), an infectious microsporidian parasite that affects the hepatopancreatic tubule cells of Penaeid shrimp. Enterocytozoon hepatopenaei does not require an intermediate host for transmission, making it a highly contagious disease with horizontal transmission. EHP infection is often characterized by slow growth and wide size distribution and can be proportional to high stocking densities (Geetha et al. 2022). It can be observed in the early stages of post-larval production in hatcheries – mainly in Thailand, Vietnam, Indonesia, Malaysia, and China. In a study by Geetha et al. (2022), EHP was shown to cause an average loss of US$813/ton of production in Penaeus vannamei shrimp farming in India.
EHP Infection in Pacific White Shrimp
In a study by Kumar et al. (2022), the digestive enzymes, metabolism, physiology, immunity, and growth responses of Pacific white shrimp (P.vannamei) at different time intervals were investigated after 90 days of EHP challenge and compared to a control group (unchallenged).
Digestive enzymes α-amylase and lipase significantly decreased (P < 0.05) in the EHP-challenged group. Metabolic variables such as triglycerides (TG), total protein (TP), cholesterol (CL), glucose (GL), and alanine aminotransferase (ALT) were significantly lower (P < 0.05) in the EHP-challenged group. Non-specific immune responses were affected by EHP infection by reducing the activity of alkaline phosphatase (ALP), catalase (CAT), γ-glutamyl transferase (GGT), total antioxidant capacity (T-AOC), superoxide anion (SOA), phenoloxidase (PO), and total hemocyte count (THC). The FCR of the challenged group was 3.01 ± 0.29, while the control group was 1.64 ± 0.05. The growth of infected animals reached only 12.17 ± 0.80 g, while the control was 19.27 ± 0.5 g after 90 days of culture.
In another study by Subash et al. (2022), Penaeus vannamei shrimp were experimentally infected with EHP by spore injection (~1x10 5 spores/shrimp) and oral feeding of infected hepatopancreas. There was a significant decrease in immune parameters total hemocyte count (THC), catalase activity (CAT), and lysozyme activity (LYS) – at 6, 24, and 24 hours post-infection (HPI), respectively.
On the other hand, EHP infection led to significantly higher levels of superoxide dismutase (SOD) activity, prophenoloxidase (proPO) activity, and respiratory burst activity (RBA) at 6 hpi. EHP infection resulted in higher oxidative stress (RBA) at 6 hpi and was counteracted by SOD and CAT to protect host cells from oxidative damage. Toll gene expression was activated as an early response when EHP interacted with the host, activating the prophenoloxidase system and the migration of hemocytes to the infection site to combat invading EHP from 6 hpi. Although appropriate responses were mounted, they appeared insufficient to eliminate EHP. Furthermore, lysozymes active from within hemocytes appeared to be neutralized by EHP or perhaps not even released from hemocytes, as hemocytes were unable to self-degenerate or phagocytose intracellular parasites when levels decreased from 24 hpi..
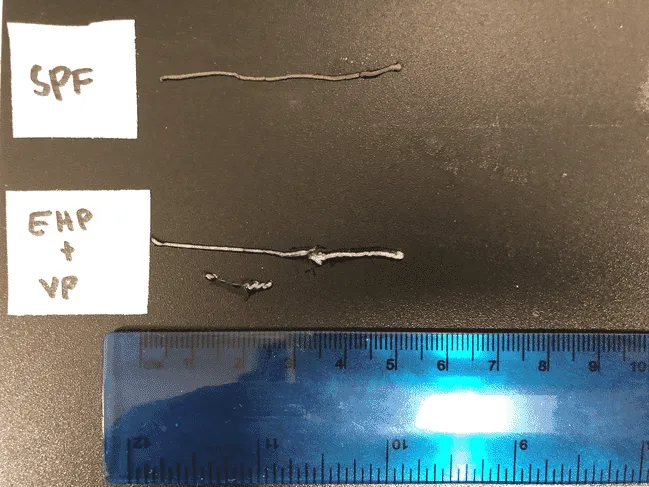
Intestines of a healthy pathogen-free shrimp (top) and one infected with EHP and Vibrio parahaemolyticus (bottom)
Cao et al. (2023) investigated changes in hepatopancreatic transcriptional levels after artificial challenge of healthy Penaeus vannamei shrimp with EHP. Differentially expressed genes (DEGs) were closely related to immune responses, while enhanced hematopoietic function and stimulation of the Jak-STAT signaling pathway led to the ultimate activation of the immune system. EHP infection increased lipid metabolism, inhibited carbohydrate and amino acid metabolism, and protein digestion – which may be the main reasons affecting normal shrimp growth.
Haemocytin was first reported in Litopenaeus vannamei shrimp (LvHCT) against EHP infection in a study by Sukonthamarn et al. (2023). Haemocytin is believed to be a key mediator of hemocyte synthesis and the prophenoloxidase (proPO) activation system. Inhibition of LvHCT resulted in higher EHP copy numbers. LvHCT plays an important role in the innate immunity of shrimp against EHP infection because LvHCT gene transcripts were downregulated after EHP infection and EHP copy numbers increased in shrimp cultured with LvHCT.
Effects of Water Quality and Farming Systems on EHP Infection
In a study by Shen et al. (2019), slow-growing small Penaeus vannamei shrimp collected from earthen ponds showed a high EHP infection rate (91.3%). Interestingly, normal-sized large shrimp in earthen ponds also had a high EHP infection rate confirmed by nested PCR (11%) and first-step PCR (72.4%). Large shrimp cultured in greenhouses had a lower EHP infection rate (10.6%) compared to normal-sized large shrimp cultured in earthen ponds (72.4%), with no EHP infection detected by first-step PCR in greenhouse-cultured shrimp. The authors suggested that greenhouse shrimp farming was less affected by EHP infection, as evidenced by normal shrimp size and weight.
Six super-intensive ponds in Indonesia were evaluated for ammonia and nitrite concentrations and EHP infection levels by Nkuba et al. (2021). A PCR test showed two ponds were positive for EHP, while nitrite and ammonia concentrations were above 1 ppm. The remaining four ponds were negative for EHP, with nitrite and ammonia concentrations below 1 ppm.
In another study, it was observed that EHP infection could occur at 2 ppt salinity with very low prevalence and severity, while infection rates and severity were higher at 30 ppt salinity (Aranguren et al. 2021).
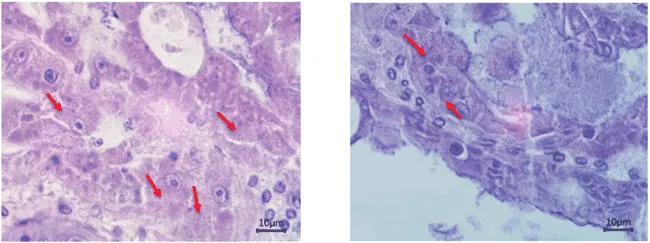
EHP-infected tissue under microscope.
Presence of EHP in other Aquatic Organisms in Shrimp Ponds
In a study by Dewagan et al. (2023) at a shrimp farm in Maoming, China, shrimp and other organisms were investigated for EHP using PCR assay. EHP was detected in Litopenaeus vannamei shrimp, Penaeus monodon, crabs, false mussels, and three dragonfly species (Anax parthenope, Pantala flavescens, and Ischnura senegalensis).
Upon histological examination, EHP spores were found in naturally infected dragonfly larvae and adults collected from shrimp ponds. In situ hybridization results showed positive signals for EHP infection in the fat body of dragonfly nymphs. Immature, mature, and late sporont plasmodium microsporidian spores were observed in the cytoplasm of dragonfly nymphs by transmission electron microscopy.
EHP transmission from shrimp to dragonfly nymphs was confirmed through challenge experiments where EHP-free dragonfly nymphs cohabited with EHP-infected shrimp. EHP transmission from dragonfly nymphs to shrimp was demonstrated through EHP-infected dragonfly larvae cohabiting with EHP-free shrimp and oral challenge trials. The study confirmed that dragonflies can be alternative hosts for EHP and that horizontal transmission of EHP can occur between dragonflies and shrimp.
In another study in Malaysia by Sajiri et al. (2023), EHP was detected in 82 aquatic samples belonging to Arthropoda, Mollusca, and Chordata phyla using PCR targeting spore wall protein (SWP) encoding genes. The average EHP prevalence by PCR was 82.93% for all three phyla (Arthropoda, Mollusca, and Chordata). These findings suggest that the presence of EHP spores in aquatic organisms in shrimp ponds are potential disease vectors (see table below).







