Results Show the Potential Benefits of Managing the Microbiome in Water and Feed Environments

The microbiome is closely related to the health status, nutrient metabolism, and immune system of farmed animals. After animals are born, their skin and gut microbiomes gradually assemble. The assembly of the microbiome in early life can have long-term impacts on host health. And there is significant interest in the relationship between the gut microbiome and the growth or health of aquatic invertebrates.
Larval production of whiteleg shrimp (Litopenaeus vannamei) is affected by various diseases, impacting larval survival and quality, which are closely related to growth, development, and resistance to stress and disease.
Currently, research on the microbiome (all microorganisms, both beneficial and harmful) of L. vannamei primarily focuses on different growth stages (larval, juvenile, or adult) regarding their association with disease outbreaks, growth, and stress resistance. Several studies have reported changes in the structure and function of the shrimp gut microbiome. Understanding how the microbiome functions in early life is based on knowing the mechanisms of microbiome inheritance and assembly.
Understanding the mechanisms of microbiome assembly can help address the debate on whether we can improve shrimp farming success rates by manipulating the microbiome. However, little is known about the dynamics and classification of bacterial assembly processes in shrimp larvae.
This article discusses the results of a study using 16S rRNA gene Amplicon sequencing (a technique used to reconstruct Phylogenies - evolutionary relationships between species, due to the slow evolutionary rate of this gene region) to investigate the inheritance and assembly processes of the bacterial community in whiteleg shrimp larvae in practical aquaculture.
Research Setup
This study was conducted in Wenchang, Hainan Province, China. Whiteleg shrimp larvae were sourced from the same broodstock pair to minimize genetic and individual differences. Larvae were cultured in 13 m3 tanks under standard shrimp production conditions, including temperatures of 30 - 32 °C; pH 8.0 - 8.3, salinity 30 - 33‰, and dissolved oxygen 5 - 8 mg/L. Shrimp were stocked at a density of 3.5 million Nauplii per tank.
Feeding occurred 6 times/day; during Zoea I - II stages, live microalgae Chaetoceros sp. and Thalassiosira sp. were fed (3 – 3 times/day). During Zoea III and Mysis stages, Artemia was fed 3 times/day and then gradually replaced with fresh feed in the subsequent stages.
Both larvae and culture water samples were collected at Nauplius, Zoea I, Zoea II, Zoea III, Mysis, and post-larval stages over 15 days (exactly 350 hours). Larval developmental stages were confirmed by microscopy. 26 sampling time points were used to collect both shrimp larvae and water samples, except for hours 31, 206, and 254 when only larval samples were taken. Sampled shrimp were processed, DNA extracted for analysis, and other laboratory procedures performed.
Results and Discussion
Results showed that the bacterial community originated primarily from the larval metacommunity (a collection of interacting communities linked by the dispersal of multiple interacting species), while planktonic microorganisms only appeared in the animal's sub-stages. This result indicates that the assembly of the bacterial community depends on inter-individual exchange, possibly through post-feeding feces or Biofloc.
Shrimp larvae (especially at the Zoea stage) often have a poor immune system and are susceptible to Zoea - II Syndrome, which can lead to high mortality rates. Several studies have shown that the diversity of the animal gut microbiome is closely related to its integrity and functional stability. Although it is argued that higher microbial diversity does not necessarily correspond to a more stable and healthier ecosystem, high diversity is generally considered to help maintain the stability and ecological function of the microbiome, thus serving as an important indicator of host health status. High microbial diversity is desirable for maintaining shrimp growth, health, and stress resistance.
The composition of the microbiome changes with host development, and a high-frequency sampling strategy facilitated identification. Both the taxonomic and species composition of the microbiome showed dependence on the larval stage, even among Zoea sub-stages. Additionally, we found distinct compositions between the water and larval microbiomes. Similar results have been previously reported for the freshwater prawn Macrobrachium nipponense.
Results showed that the bacterial family Rhodobacteraceae was dominant compared to other bacterial groups. There were changes in the bacterial community, evidenced by strong fluctuations in the relative abundance of Rhodobacteraceae, Cyclobacteriaceae, and Flavobacteriaceae (Chart 1). This could be due to differences in the environmental conditions of the hatchery systems leading to differences in the larval microbiome.
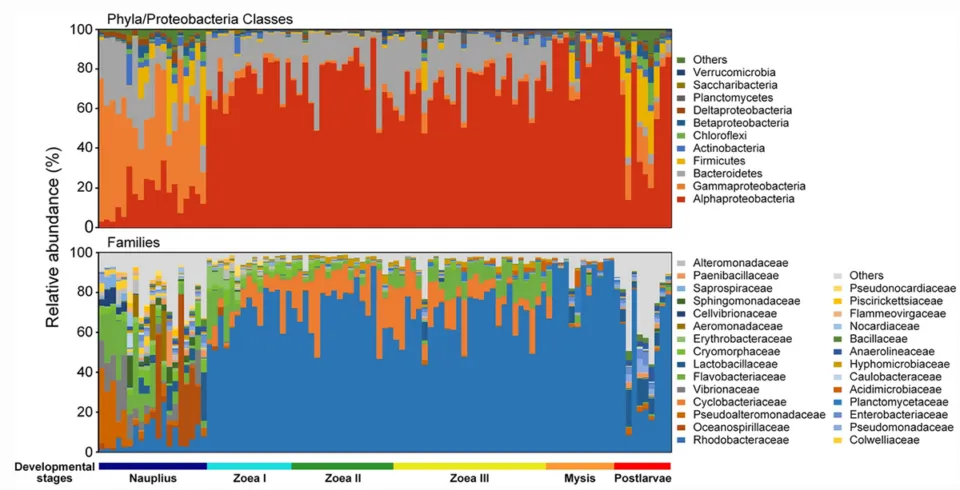
Chart 1. Composition of the bacterial community in whiteleg shrimp larvae
The study showed that changes in physiological state and nutritional regime can lead to significant differences in the larval bacterial community. Many studies have found Rhodobacteraceae bacteria to be dominant in the shrimp gut, suggesting it may be a core gut microbial group of whiteleg shrimp.
Rhodobacteraceae are heterotrophic bacteria with high diversity and flexibility in organic matter degradation, and are widely distributed across various marine ecosystems. And most taxa belonging to the family Rhodobacteraceae can synthesize Vitamin B12, an essential nutrient for shrimp. Furthermore, this family can significantly contribute to the biosynthesis and metabolism potential of various organic compounds, participating in the conversion of organic matter in the larval digestive tract and providing essential nutrients for host development.
Additionally, the abundance of Rhodobacteraceae in the gut bacterial community of healthy individuals is often higher than in diseased individuals and shows an antagonistic relationship with potential pathogens such as Vibrio. And the family Rhodobacteraceae could be a potential source of Probiotics for larval rearing.
Source: www.aquaculturealliance.org
Abridged translation by: Trần Thị Thúy Quyên







