Currently, crustaceans hold an important economic position in global aquaculture, with black tiger shrimp (Penaeus monodon) and whiteleg shrimp (Litopenaeus vannamei) being the two main cultured species. However, shrimp farming always faces inherent risks and diseases, with the greatest losses still caused by Vibrio and viral diseases.
Information on β-glucan compounds extracted from seaweed and their mechanism of stimulating the non-specific immune system in crustaceans is presented as a basis for research on extracting β-glucan compounds from several seaweed species found in the coastal areas of the Mekong Delta, contributing to the development of sustainable shrimp farming.
Betaglucan – an abundant resource in the ocean
β-glucan is a Polysaccharide (PS) composed of monosaccharides. The linkage positions of monosaccharides in the chain form different compounds: Agar (β-1,3-1,4-glucan), Alginate (β-1,4 glucan), Carrageenan (β-1,3-1,4-glucan), Fucoidan (β-1,3-glucan), Laminarin (β-1,3-1,6-glucan), etc. Agar and Carrageenan are mainly extracted from red algae species (Rhodophyta), while Fucoidan, Laminarin, and Alginate are abundant in brown algae species (Phaeophyta). Additionally, other forms of β-glucan such as Chrysolaminarin (β-1,3-1,6-glucan) are rich in microalgae, and Zymosan (β-1,3-glucan) is found in yeast (Figure 1).
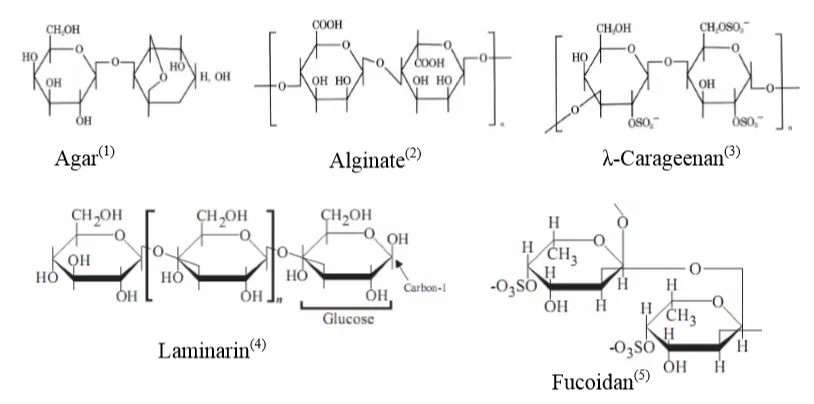
Figure 1. Structure of Betaglucan compounds
PS are usually extracted with water or solvents, forming neutral mixtures or acidic high molecular weight compounds. PS extracted from seaweed were first described as having β-glucan-like activity. In the ocean, forms of β-glucan are also found in the cell walls of fungi and bacteria.
Biological activity of Betaglucan extracted from seaweed
The cell walls of some marine algae species contain Fucoidan compounds, which are not found in terrestrial plants and have special functions in ion regulation. Furthermore, these compounds are used as antioxidants, anticoagulants, anti-cancer, anti-inflammatory agents, and immune stimulants.
Mechanism of Betaglucan immune stimulation
Shrimp do not possess a specific immune system; their immune response against pathogens relies solely on innate immunity, including physical barriers (chitinous shell), humoral immunity, and cellular immunity.
Crustacean hemocytes are divided into two types: hyaline cells and granular cells. Cerenius and Söderhäll (2004) suggested that β-glucan stimulates melanization and phagocytosis through recognition by specific pattern recognition proteins (PRPs): LGBP (lipopolysaccharide and β-1,3-glucan-binding protein) and βGBP (β-glucan-binding protein) on hemocytes (Figure 2).
When granular hemocytes are stimulated by β-glucan, degranulation occurs, leading to the release of several immune enzymes: Prophenoloxidase, Serine Proteinase, Peroxinectin, and α2-macroglobulin. Subsequently, with the catalysis of Trypsin-like serine proteinase (SP), the proPO system is activated from its inactive form to active Phenoloxidase (PO). PO is a Cu-containing enzyme that further catalyzes two processes: O-hydroxylation of Monophenol and oxidation of Diphenol into Quinone. An important function of PO is to convert Tyrosine into Dihydroxyphenylalanine (DOPA) as well as DOPAquinone. During the melanization process, Melanin encapsulates bacteria/foreign bodies and is released outside the chitinous shell.
While melanization occurs in granular hemocytes, phagocytosis occurs in hyaline cells. During this process, atomic oxygen radicals (O-), hydroxyl radicals (OH-), and Hydrogen Peroxide (H2O2) are generated and are considered potent oxidants that kill bacteria. Consequently, the activities of SuperOxide Dismutase (SOD), Gluthathione Peroxidase (GPx), and Catalase (CAT) enzymes also increase to balance the strong oxidizing radicals produced by hemocyte phagocytosis.
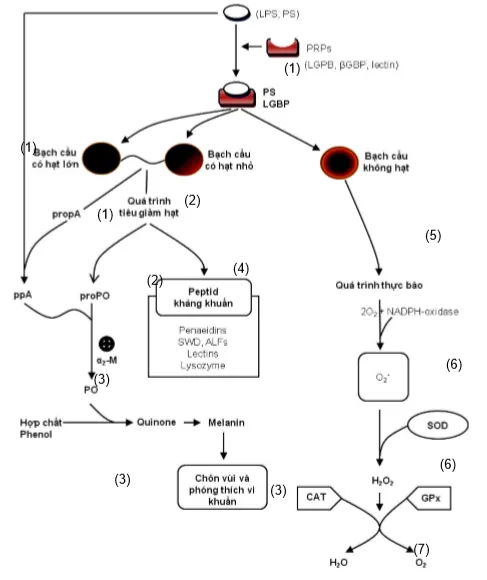
Figure 2. Mechanism of non-specific immune stimulation in shrimp when stimulated by Betaglucan
Applications and Prospects
Regarding β-glucan compounds extracted from seaweed, Chotigeat et al. (2004) also conducted research on the brown alga Sargassum polycystum. Crude Fucoidan from S. polycystum was extracted using 0.1 N HCl solution. The experiment was performed on black tiger shrimp of two different sizes (5–8 g and 12–15 g). Results showed that shrimp weighing 5–8 g fed at a dose of 400 mg/kg shrimp/day increased survival by 46% after 10 days of white spot virus infection. For shrimp weighing 12–15 g, a dose of 200 mg/kg shrimp/day increased survival up to 93% after 11 days of infection, and the phagocytic index reached 2.36 ± 1.28 compared to the control's 0.83 ± 0.6. The authors also indicated that crude Fucoidan extracted from the brown alga S. polycystum has antibacterial properties, with minimum inhibitory concentrations (MIC) against E.coli, Staphylococcus aureus, and Vibrio harveyi being 6.0, 12.0, and 12.0 mg/mL, respectively.
Hou and Chen (2005) also studied the application of a mixture extracted from the red alga Gracilaria tenuistipitata to stimulate the immune system in whiteleg shrimp through injection at a dose of 4–6 µg/g shrimp. Results showed that 2 hours after injection, the total hemocyte count (THC), PO activity, Super anion O2x- (RB), and survival rate of the experimental shrimp significantly increased (p< 0.05) compared to the control group.
On whiteleg shrimp, Yeh et al. (2006) also investigated the immune-stimulating ability of a mixture extracted from the brown alga S. duplicatum. Shrimp were immersed in a solution of 300–500 mg/L of the extract mixture and injected at a dose of 10–20 µg/g shrimp, which increased immune parameters (THC, PO, and RB) after 1 hour of immersion and 1 day of injection, respectively. Phagocytic activity also significantly increased (p< 0.05) in these treatments when shrimp were injected with V. alginolyticus at a dose of 1.8 × 106 CFU for both immersion and injection experiments.
On Indian white shrimp, Huang et al. (2006) reported that compounds extracted from the alga S. fusiforme helped shrimp enhance immunity and resist V. harveyi bacteria. Indeed, PO and Lysozyme enzyme activities increased up to 13.2 and 3.21 units/mg protein when shrimp were fed diets supplemented with the extract mixture at a dose of 0.5%. The study results also demonstrated that doses of 0.5–1% of the diet increased survival rates when shrimp were challenged with V. harveyi at a dose of 9.3 × 107 CFU after 14 days of experimentation.
In summary, research on the application of β-glucan extracted from seaweed has been limited to laboratory scale. Currently, there are no studies on the application of seaweed extracts as supplements in commercial shrimp farming, especially in intensive farming.
The feeding method is considered highly feasible, non-stressful, and applicable at all stages of shrimp development. Therefore, research on supplementing shrimp feed with β-glucan extracted from seaweed to enhance shrimp resistance is crucial. Natural seaweed resources are abundant, providing a large amount of β-glucan at low cost. Thus, successful research on extracting β-glucan compounds from seaweed will increase the economic efficiency of shrimp farming.
Source: Huỳnh Trường Giang, Vũ Ngọc Út and Trương Quốc Phú – Can Tho University







