Elevated hydrogen sulfide (H2S) concentrations often lead to lethal effects on fish. This adverse effect manifests at different concentrations due to various parameters, such as temperature, oxygen saturation, and salinity, as well as redox potential, which influence the toxicity of H2S in water. The presence of oxygen acts as a protective agent against H2S through oxidation, but this process is too slow to protect fish in cases of sudden H2S release into the environment.
Anaerobic environments with H2S can form anywhere in a fish farm, especially in biofilters with lethal concentrations when high, insufficient aeration, and stagnant piping systems. Sulfate (SO2) concentrations in seawater (2,700 mg/L) are significantly higher than in freshwater (5-50 mg/L) (Letelier-Gordo et al., 2020). However, a fish farm needs to operate at a salinity below 5ppt before SO2 becomes limiting for H2S formation.
Sulfate-reducing bacteria (SRB) compete with nitrate-reducing bacteria (NRB) for carbon in the form of volatile fatty acids. If a fish farm operates at higher nitrate levels, NRBs can outcompete SRBs for limiting volatile fatty acids. Some evidence even demonstrates its use as a remedy for H2S accumulation rather than a preventative measure in shrimp ponds (Torun et al., 2020). However, this solution presents issues with less environmentally sustainable nitrogen discharge.
Different fish species have varying sensitivities to H2S in water. Literature indicates that fish are highly sensitive to H2S. Levels of 2µg/L in freshwater and 5µg/L in saltwater can cause acute stress and even mortality after 2-4 weeks of chronic exposure, according to a newer, unpublished study (Nofima, 2022). Some literature suggests that concentrations above 25µg/L can be lethal to fish. Recent research proposes a limiting H2S concentration of 60.7 ± 13.2 µg/L at 14°C (Bergstedt & Skov, 2023). However, there is no final consensus on H2S concentrations.
Biofilters have been reported to produce up to 139g of H2S per m³ of biofilter volume when mixing and aeration are insufficient. Therefore, it is extremely important to monitor biofilter performance (Rojas-Tirado et al., 2021).
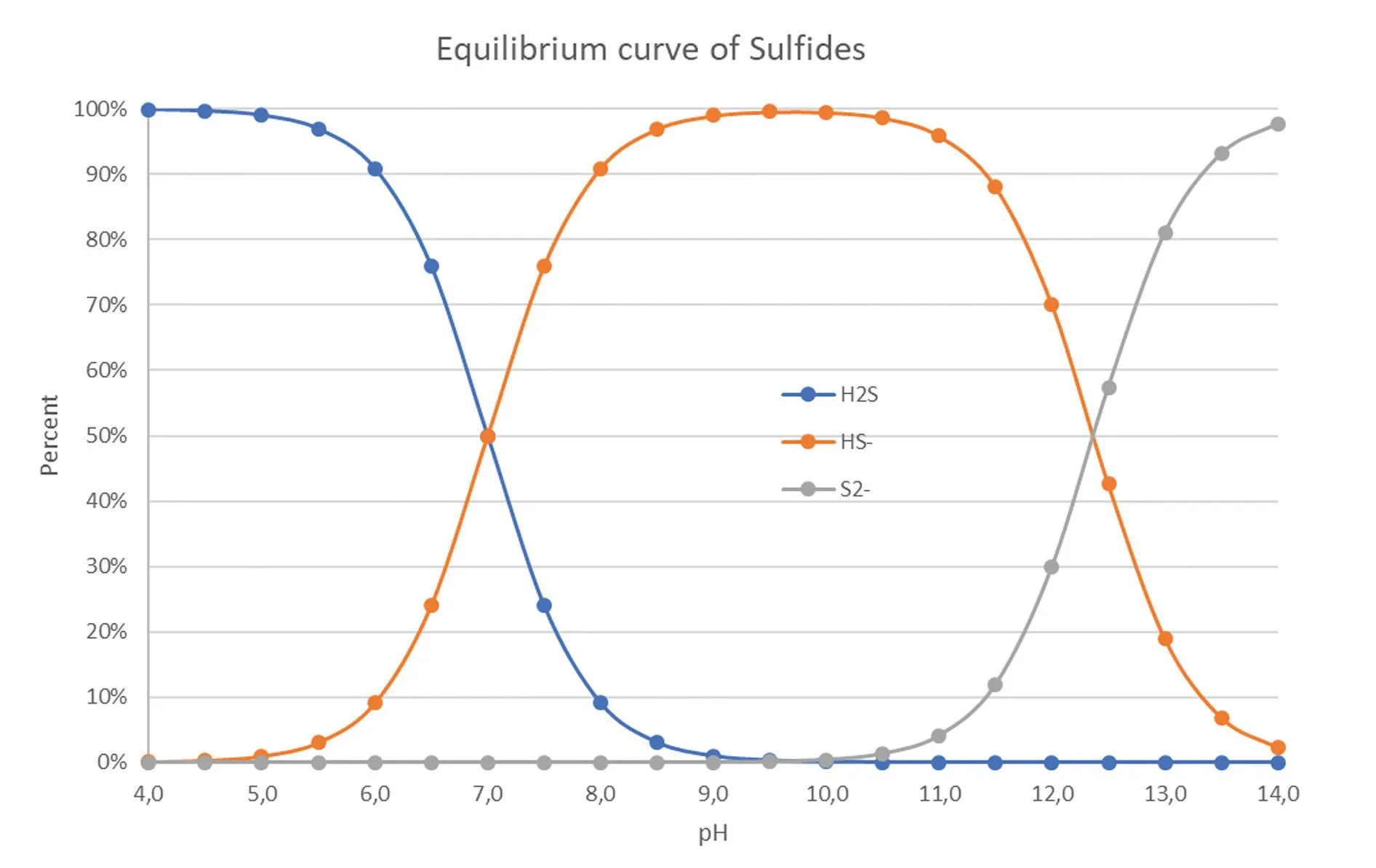
The relative concentration of H2S is most dangerous at lower pH values. Conversely, it becomes less dangerous at higher pH values, where it exists as H2S. Gaseous H2S readily binds with water and exists in equilibrium in its aqueous form, with approximately 72% existing as H2S and 28% as aqueous H2S solution at pH 7.4 (Li & Lancaster, 2013).

Continuous removal and monitoring of hydrogen sulfide H2S at fish farms. H2S binds with metals to form metal sulfides. H2S can be reduced by chemical oxidation and oxidation by ozone, peroxide, or in biological oxidation processes with nitrate by bacteria (Bergstedt et al., 2022). Chemical oxidation via oxygen or peroxide has proven particularly effective in aquaculture as this method is commonly used for disinfection or oxidation. This targeted approach often proves highly effective by targeting the larger H2S group rather than the significantly smaller H2S group at operational pH levels (Bergstedt et al., 2022; Bögner et al., 2021).
Fish farms should minimize H2S accumulation. Today, there are systems that can measure total H2S multiple times a day at various locations across a RAS, generating up to 72 daily data points. This technology effectively measures H2S and HS in water samples by lowering the pH of the water sample. This yields higher gaseous H2S results, which can be extracted by running the more acidic water sample through a dual membrane system. This allows for earlier detection of concentration changes compared to other systems that only measure H2S alone, as the detection limit is lowered, potentially leading to inaccurate results.
Monitoring H2S to microgram levels by itself is not the most important protective measure against sudden H2S release. A well-documented issue is that feces and residual organic matter provide the basis for H2S release in recirculating systems (Rojas-Tirado et al., 2021). Therefore, understanding the process of fecal removal and the quality of biofilters is crucial for protection against H2S in recirculating systems.
Although fish feed is currently often optimized to produce compact feces, the hydrodynamics within the fish tank will often grind feces into smaller particles before they can be effectively removed mechanically. Data indicates that some fish tank designs lead to dissolved feces forming 'sludge,' which increases the risk of H2S release and requires complete cleaning using expensive methods.
By monitoring turbidity and particle size before and after the fish tank, managers can directly track how many fecal particles are being broken down before mechanical filtration. By monitoring turbidity and particle size on both sides of the mechanical filter, managers can know how many particles are escaping mechanical removal, with finer particles escaping.
'Turbidity measures the interaction of light with suspended particles of varying sizes, shapes, colors, and reflectivities. A particle counter quantifies both particle size and number and is potentially an even more useful tool.

Mechanical removal often gets stuck inside the biofilter. Again, by monitoring both sides of the biofilter, managers can track the quantity and size of particles accumulated or released over time. This provides a rich picture of both the biofilter's quality and the overall farm's quality.
The most effective way to monitor these gradients and manage the performance of fish tanks and filters is by using advanced centralized measurement technologies. This also helps reduce the cost of purchasing multiple pieces of equipment on the farm because it allows single high-end sensors to monitor multiple locations on the farm instead of investing in many lower-end sensors for the same cost. Installing multiple sensors also comes with high technician costs for proper installation. Companies offering turnkey data sampling solutions often have a linked cloud solution so customers can view their online data in real-time or near real-time from anywhere in the world on a computer, tablet, or even a phone.
However, these technologies can be used with many additional parameters. For example, by monitoring total inorganic carbon, pH, oxygen, and ammonia, managers can better manage a biofilter with fewer anaerobic zones that could produce H2S.

Caspar Yan Hansen
Data & Analytics Consultant, Blue Unit AS
E: cyh@blue-unit.com

David Owen
Head of Technology, Blue Unit AS
E: dowen@blue-unit.com






