Problem Statement
The Bagridae family is one of the largest fish families in Asia. Species within the Bagridae family are of particular interest due to many species having ornamental value compared to other Asian catfish species. There have been several domestic and international studies on the artificial reproduction of small-sized, ornamental Bagridae species, mainly belonging to the genus Mystus. However, to date, there have been no studies on the induced breeding and artificial propagation of Striped Dwarf Catfish.
Research to evaluate the artificial reproduction potential of Striped Dwarf Catfish under captive conditions using different types of reproductive stimulants is essential. The research results will contribute to establishing an artificial propagation protocol and developing Striped Dwarf Catfish farming, thereby diversifying aquaculture species and restoring natural populations of this species.
Materials and Methods
RESEARCH MATERIALS
Broodstock selected for conditioning and breeding had characteristic appearance and coloration, were healthy, and met size requirements (total length > 11 cm, weight 19 - 25 g/individual). Fish were conditioned in earthen ponds with trash fish feed until sexual maturity.
Three types of reproductive stimulants (CKTSS) commonly used in artificial fish reproduction are carp pituitary gland (FPG) preserved in acetone, human chorionic gonadotropin (HCG), and gonadotropin-releasing hormone analog (LHRHa).
EXPERIMENTS
Experiment 1: Evaluating the reproductive stimulation potential of different reproductive stimulants on Striped Dwarf Catfish
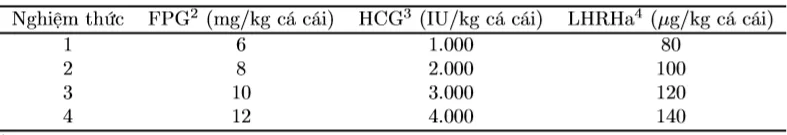
Experiment 1 was conducted with different CKTSS (FPG, HCG, and LHRHa), applying a two-dose injection method with the same type of CKTSS for female fish. The time interval between the priming dose and the resolving dose injection was 8 hours. Male fish were injected with the same type of CKTSS as females, with a single injection administered simultaneously with the resolving dose for females. The priming dose was 2 mg/kg FPG, 500 IU/kg HCG, and 40 µg/kg LHRHa, and the dose for male fish was approximately 1/3 of the total dose for female fish. The number of treatments (NT) and the dosage of the resolving injection for female fish are presented in Table 1. In each treatment, 10 female fish were used, with 3 replicates. After CKTSS injection, fish were kept in glass tanks (0.8 × 0.5 × 0.5 m) with gentle aeration.
Table 1. Number of treatments and CKTSS doses in the experiment evaluating the reproductive stimulation potential of Striped Dwarf Catfish
After 10 hours post-resolving dose injection and being maintained at a water temperature of 28°C, female fish from the same treatment and 1 replicate were stripped into a clean dish. All eggs were inseminated with milt from 1 male fish using the semi-dry fertilization method. After fertilization, eggs were adhered to a nylon mesh substrate and incubated in a Weis jar with continuous water flow. In each fertilization batch, approximately 200 eggs were kept in an aerated glass jar to calculate fertilization rate, hatching rate, and larval survival rate.
Experiment 2: Determining the appropriate timing for stripping eggs and artificial insemination using different reproductive stimulants
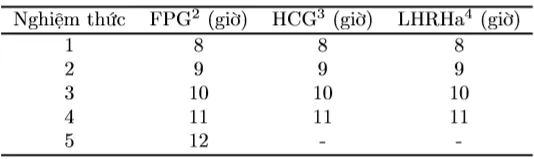
Experiment 2 was conducted with different CKTSS (FPG, HCG, and LHRHa). The CKTSS injection method for broodstock in Experiment 2 was the same as in Experiment 1. The resolving dose for female fish was the optimal dose determined in the experiment evaluating the reproductive stimulation potential of Striped Dwarf Catfish, and the dose for male fish was approximately 1/3 of the total dose for female fish. The number of treatments and the timing for stripping eggs and artificial insemination are presented in Table 2. In each treatment, 10 female fish were used, with 3 replicates. After CKTSS injection, fish were kept in glass tanks with gentle aeration.
Table 2. Number of treatments and timing for stripping eggs and artificial insemination for Striped Dwarf Catfish using different CKTSS
After the resolving dose injection and being maintained at a water temperature of 28°C, at specific time points post-resolving dose injection, female fish from the same treatment and 1 replicate were stripped into a clean dish, and artificial insemination was performed as in Experiment 1. The sampling method for calculating fertilization rate, hatching rate, and larval survival rate was also the same as in Experiment 1.
In these experiments, the measured environmental parameters for the spawning tanks, incubation tanks, and nursery tanks were as follows: temperature 28 - 31°C, pH = 7, and DO > 4 mg/L. These environmental factors were suitable for broodstock reproduction and the development of Striped Dwarf Catfish embryos and larvae.
Research Results
REPRODUCTIVE STIMULATION POTENTIAL OF REPRODUCTIVE STIMULANTS
Table 3. Reproductive parameters of Striped Dwarf Catfish induced to spawn with FPG, HCG, and LHRHa
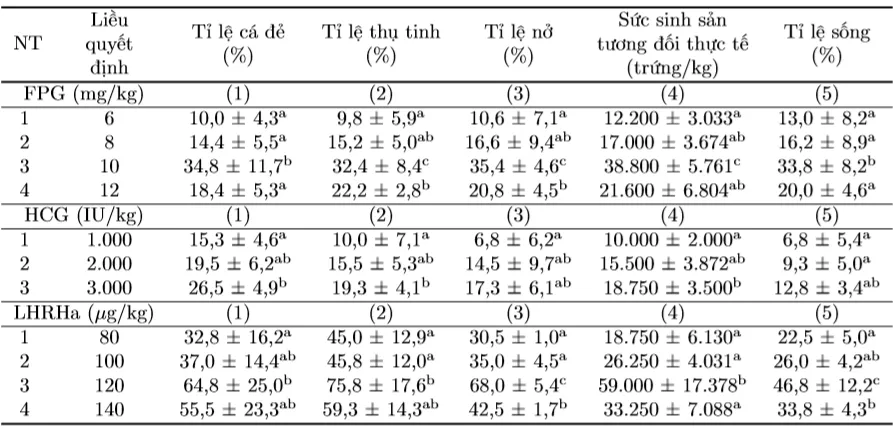
The results showed that the optimal resolving dose for FPG was 10 mg/kg female fish, for HCG was 4,000 IU/kg female fish, and for LHRHa was 120 µg/kg female fish.
APPROPRIATE TIMING FOR EGG STRIPPING AND ARTIFICIAL INSEMINATION
Table 4. Reproductive parameters of Striped Dwarf Catfish at different egg stripping and artificial insemination timings when induced to spawn with FPG, HCG, and LHRHa
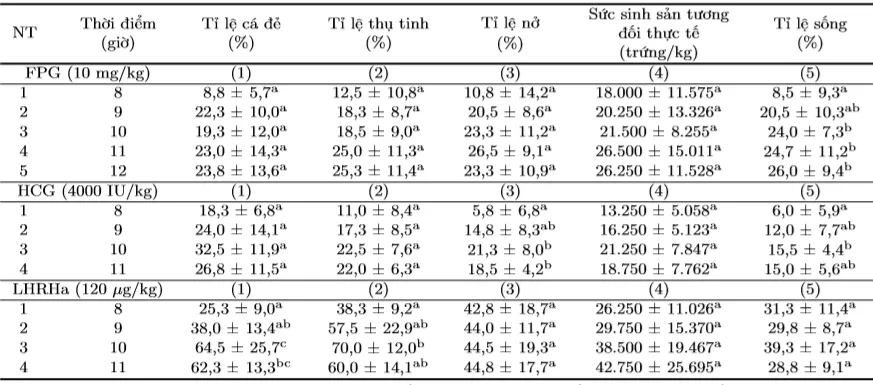
The results showed that the appropriate timing for egg stripping and artificial insemination when inducing Striped Dwarf Catfish to spawn with FPG could be from 9 hours, with HCG 9 - 10 hours, and with LHRHa 10 - 11 hours.
EMBRYONIC DEVELOPMENT
Swollen Striped Dwarf Catfish eggs had an average diameter of 1.11 ± 0.05 mm. The embryonic development time from fertilization to hatching at 28 - 30°C was 19 - 21 hours.
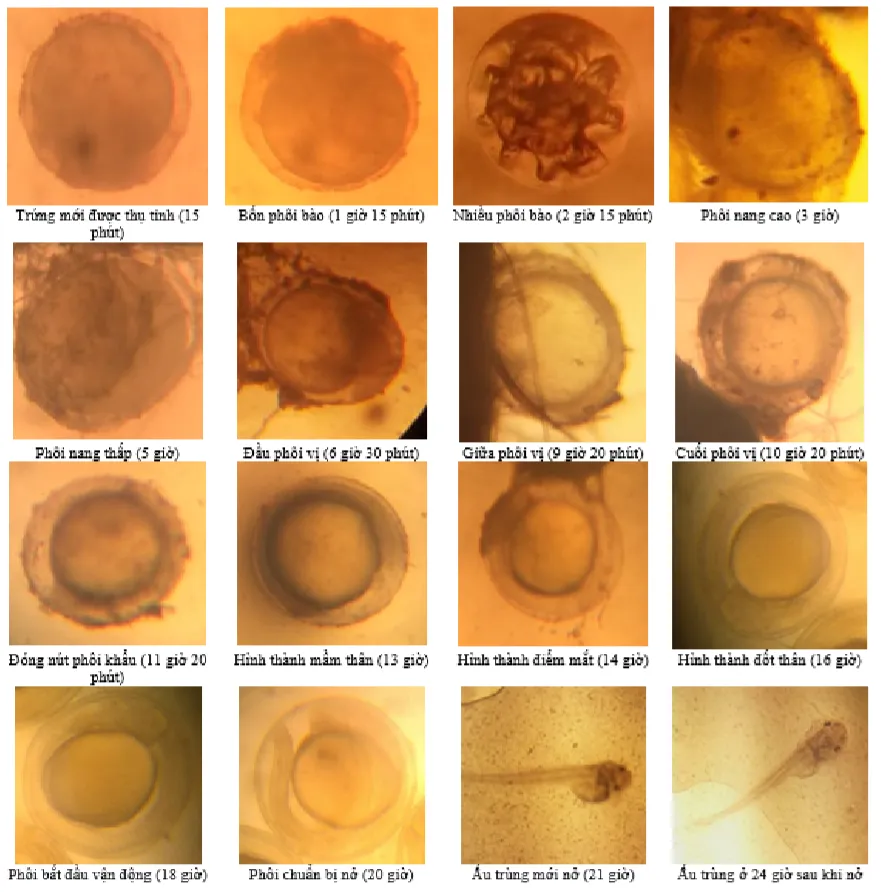
Figure 1. Embryonic development stages of Striped Dwarf Catfish (embryo and larval sizes not to scale)
Conclusion
With a two-dose injection method, FPG, HCG, and LHRHa all induced ovulation and stimulated Striped Dwarf Catfish reproduction, with optimal resolving doses of FPG at 10 mg/kg, HCG at 4,000 IU/kg, and LHRHa at 120 µg/kg female fish. LHRHa was a more suitable CKTSS for inducing artificial reproduction in Striped Dwarf Catfish compared to FPG and HCG.
The appropriate timing for stripping eggs and artificial insemination when inducing Striped Dwarf Catfish to spawn was from 9 hours with FPG, 9 - 10 hours with HCG, and 10 - 11 hours with LHRHa.
Striped Dwarf Catfish belong to a group that is difficult to induce to spawn. A dose of 120 µg/kg female fish of LHRHa and egg stripping and insemination timing of 10 - 11 hours are suitable for achieving the highest results in artificial reproduction of Striped Dwarf Catfish.
A reliable method needs to be developed to assess the reproductive readiness of Striped Dwarf Catfish before applying CKTSS to improve reproductive outcomes.
Source: Faculty of Fisheries – Nong Lam University, Ho Chi Minh City






