Understanding changes in water quality and bottom quality helps manage risks effectively
In this article (edited and summarized from the original publication in Revista Acuacultura - Cámara Nacional de Acuacultura, No. 122, April 2018), we will explain how heavy rains directly and indirectly affect shrimp ponds, how their impacts are interrelated, how they affect shrimp health, and what shrimp farmers can do to mitigate losses associated with this climatic phenomenon.
The latest forecasts from the U.S. National Oceanic and Atmospheric Administration (NOAA) indicate a prolonged period of the typical "La Niña" phenomenon, such as droughts and low temperatures worldwide (Ecuador), which will lead to continued heavy rainy seasons annually. The question arises: What do these rains mean, and how do they affect shrimp ponds?
Potentially, these rains can cause significant damage to farmed shrimp, although several other factors (unrelated to this climatic factor) influence shrimp mortality rates in ponds. In both parts of the South Pacific, farmed shrimp mortality rates have been reported to range from 3% to 50% within 3-4 days of heavy rainfall.
Initial disease symptoms are often undetectable. Typical signs are also very difficult to describe impending problems.
Rainfall directly affects physical factors and indirectly affects the pond ecosystem. Rain not only impacts the chemical factors of shrimp ponds but also significantly alters the ecological balance of the pond for an extended period.
Direct Effects of Rain
Rain typically has a temperature 50C to 60C lower than the pond environment; some areas may be even lower. Due to the dissolution of carbon dioxide (CO2), rain contains a certain amount of carbonic acid with a pH ranging from 6.2 to 6.4 (in non-industrial areas). These two physical factors tend to reduce the temperature and pH of shrimp ponds. Additionally, due to the dilution by rainwater, salinity and hardness also decrease due to the reduction in ion concentration in the environment.
Rain will directly change physical factors including: an increase in total suspended solids (TSS) due to runoff from soil and banks into the pond. Increased turbidity due to rain reduces sunlight penetration and causes a sudden disturbance to the population of photosynthetic bacteria (Phototrophic Bacteria).
The formation of a surface water layer (halocline) in the pond due to heavy rain changes the salinity in the pond.
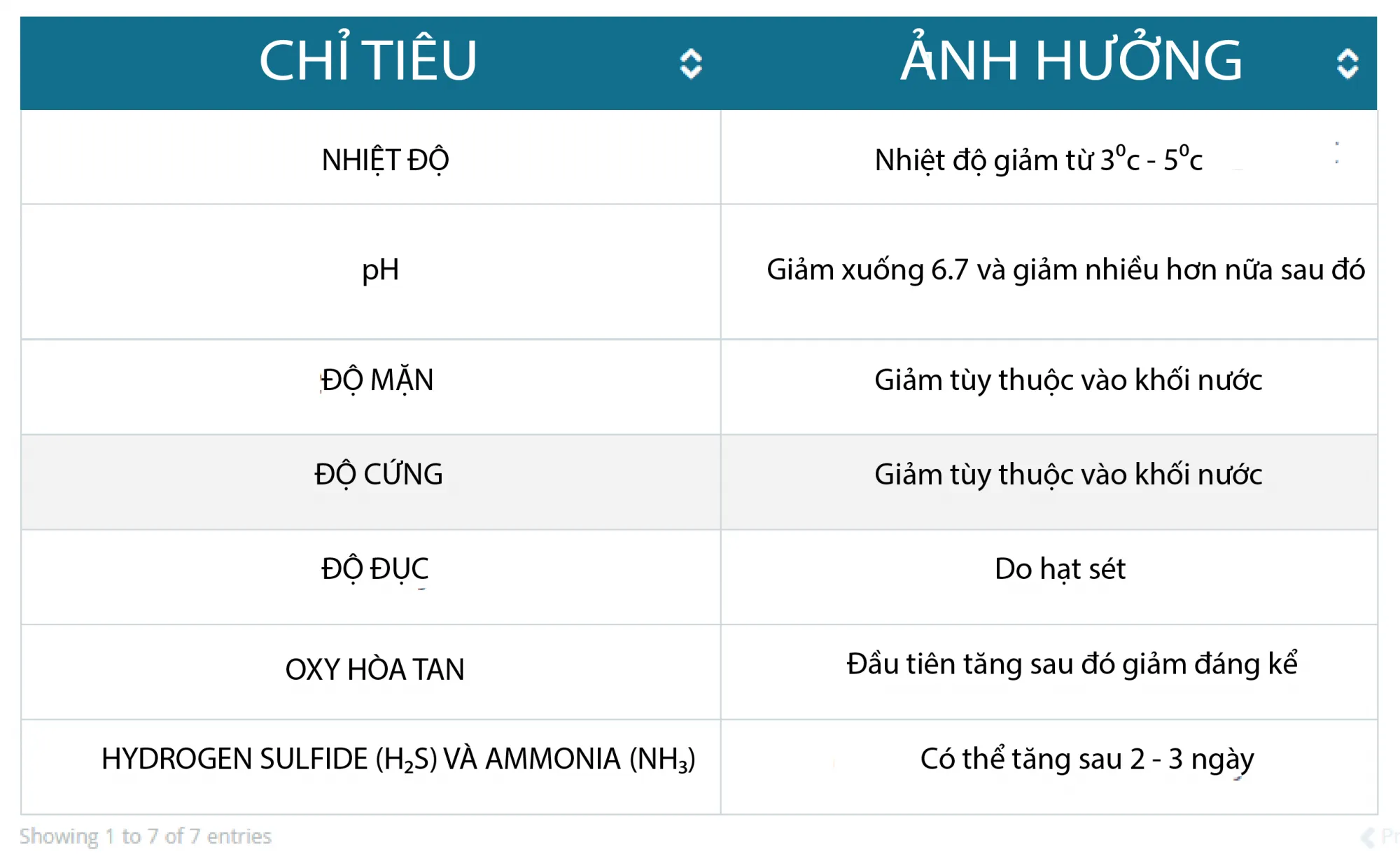
Table 1. Effects of rainfall on water quality parameters (According to Buike)
Indirect Effects of Rain
A chain of events is altered by the onset of rains. A decline in microalgae populations occurs just after (or during) the rains. This leads to a drop in pH (rainwater has a relatively acidic content), reduced mineral and micronutrient concentrations, increased turbidity, decreased light intensity, etc. Subsequently, heterotrophic bacterial populations – which play a role in decomposing organic matter – multiply exponentially due to the increase in nutrients from dead algal cells settling at the pond bottom.
At this point, dissolved oxygen (DO) levels continuously decrease. High biological oxygen demand (BOD) by heterotrophic bacteria and CO2 produced by autotrophs (which have died) lead to oxygen deficiency in a very short time if no timely remedial measures are taken. In addition to consuming available oxygen, bacterial respiration produces CO2 dissolved in the water, which will further reduce pH. Consequently, low DO, pH, and temperature create a very unfavorable environment for shrimp farming. Under these conditions, along with a large amount of organic matter, it is an ideal environment for the rapid proliferation of bacteria. Specifically, the Vibrio spp. group often predominates under these conditions, acting as a pathogen in farmed shrimp.
And under these conditions, the redox potential of the sediment may silently decrease. All compounds will be reduced under these conditions. Under reducing conditions and low pH, hydrogen sulfide (H2S) is extremely toxic to crustaceans at concentrations that would normally not be problematic. Hydrogen sulfide (H2S) is highly toxic due to its interference with oxidation and aerobic respiration. This effect is limited to H2S gas, as the HS- ion does not exhibit toxicity under normal farming conditions.
Impact on Shrimp Health
Temperature
Environmental temperature greatly influences the metabolic rate of all poikilothermic organisms (whose body temperature varies similarly to the ambient temperature), and shrimp are such organisms. Typically, feed consumption decreases by about 10% (dry weight) when the temperature drops by 10C. Because rain often lowers water temperature by 30C to 50C, this results in a minimum 30% reduction in feed consumption.
Rain will cause water stratification in the pond. The surface water layer should be immediately removed by mechanical methods to avoid significant temperature changes in the environment.
In addition to reduced shrimp feeding, temperature stratification will cause shrimp to move to areas of the pond with higher temperature and salinity, and areas that may be away from the sound of rain on the pond surface. This leads to a significant increase in shrimp density in some deeper pond areas, where dissolved oxygen concentrations are lowest and toxic gas concentrations are relatively high. If the normal feed amount is still applied, it will lead to excess feed, and bacteria decomposing the excess feed will exacerbate the pond conditions due to decreased pH and increased BOD from the aerobic respiration of heterotrophic bacterial populations.
Acidity (pH)
Rainfall has a pH of 6.5 - 6.7 in coastal Ecuador, and shrimp ponds typically have pH values of 7.5 - 8.5, with a decrease of 0.3 - 1.5 often occurring during the rainy season. The sudden destruction of the phytoplankton population in the pond occurs; it is important to note that reduced salinity leads to many other problems in the pond, with cyanobacteria dominating under low salinity conditions.
The mortality of phytoplankton provides a large amount of simple sugars to the pond ecosystem because autolysis breaks down cell walls and releases cytoplasm into the water. Within a few hours, there is often an exponential increase in heterotrophic bacteria that begin to assimilate these sugars. The energy for soil exchange requires a lot of oxygen before transitioning to an anaerobic phase, reducing metabolism. Aerobic respiration also produces CO2, which upon hydrolysis forms carbonic acid, subsequently lowering the pH. Therefore, a short-term pH disruption caused by rains can lead to serious problems, where the pH will continue to decrease until phytoplankton populations can recover.
Dissolved Oxygen
Dissolved oxygen (DO) levels are the most critical







