Diseases in pangasius are considered one of the significant challenges in the aquaculture industry. Besides the most common diseases such as: hemorrhage, liver pus, white liver, white gills, white skin, fin rot, and jaundice... Recently, swim bladder swelling in pangasius has appeared with high frequency, causing losses to farmers. Although there have been some studies on fish swim bladder diseases such as: carp with swim bladder inflammation (Markiewicz, 1966 cited by Roberts, 2000) and fungal infection of Phialophora spp. on Atlantic salmon swim bladder (Lumsden, 2006). However, "swim bladder distension" in pangasius is a new issue. In Vietnam, there are currently no studies on this disease or methods for its prevention and treatment.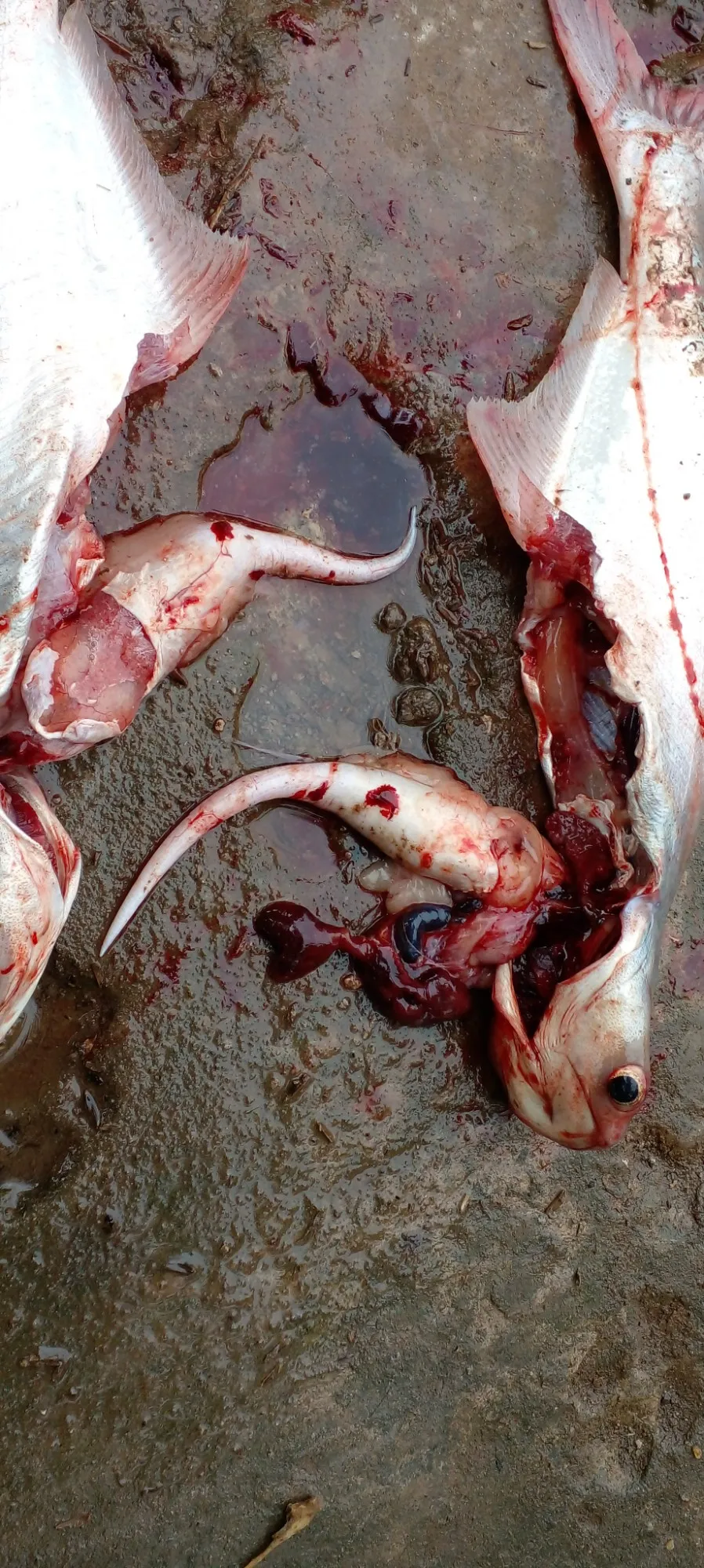
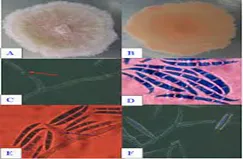
Fresh samples were observed under a microscope, and fungi were isolated on GYA medium. Isolated samples were incubated at 280C for 1-4 days. Based on the morphological characteristics of colonies, hyphae, conidiophores, and spores. The isolation results identified the fungal genus Fusarium sp. on diseased fish as Fusarium oxysporum and Fusarium subglutinans. The results also confirmed that these are the main causative agents of the disease.
However, in current high-density farming environments, we often overlook the dynamics of dissolved oxygen in the water. Dissolved oxygen in water primarily comes from diffusion from the air, especially in flowing water bodies. The dissolution of oxygen also follows Henry's Law and can be calculated using the following formula:
Cs = Ks x P
Where: Cs = gas solubility
Ks = solubility coefficient
P = partial pressure of gas
For example, at 300C and 1 atm (760mm Hg), dissolved oxygen content = 26.1 mL/L x 0.209 = 5.5 mL/L or = 5.5 mL/L x 1.4 = 7.7 mg/L (32,000mg/22,400 mL = 1.4). The percentage saturation of oxygen in water depends on pressure, temperature, and salinity. Water dissolving more or less than the saturation concentration is called supersaturated or undersaturated. The phenomenon of supersaturated dissolved oxygen often occurs due to changes in temperature and pressure. Dissolved oxygen in water also results from photosynthesis by aquatic plants, a process that often occurs strongly in stagnant water bodies.
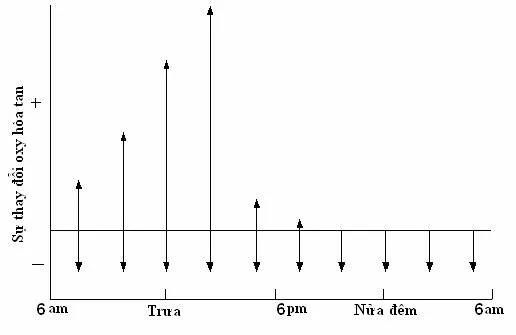 The increase (photosynthesis) and decrease (respiration) of dissolved oxygen content in nutrient-rich fish ponds.
The increase (photosynthesis) and decrease (respiration) of dissolved oxygen content in nutrient-rich fish ponds.In aquaculture ponds, oxygen levels fluctuate significantly throughout the day and night, with the extent of fluctuation depending on nutrient levels and plant growth. In nutrient-poor ponds, plants are underdeveloped, so the oxygen fluctuation range is small.
In nutrient-rich ponds, plants grow vigorously during the day, and their photosynthesis causes dissolved oxygen levels to rise significantly above saturation, reaching their peak around 14:00-16:00. Conversely, at night, the respiration of aquatic organisms consumes a lot of oxygen, causing dissolved oxygen levels to gradually decrease and reach their lowest point in the early morning. In overly nutrient-rich ponds, dissolved oxygen levels in the early morning can drop to 0 mg/L and reach supersaturation of 200% by midday.
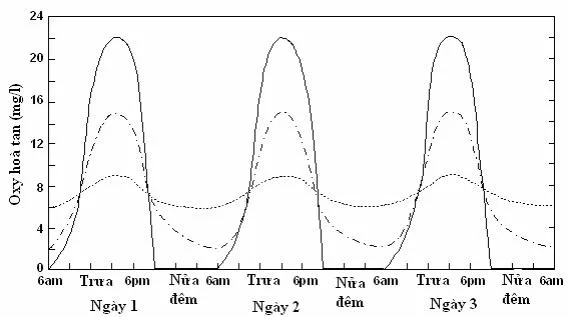 Day and night changes in dissolved oxygen content (mg/L) in nutrient-poor ponds (dotted line), nutrient-rich ponds (dashed line), and overly nutrient-rich ponds (solid line).
Day and night changes in dissolved oxygen content (mg/L) in nutrient-poor ponds (dotted line), nutrient-rich ponds (dashed line), and overly nutrient-rich ponds (solid line).In an aquaculture pond, nutrient levels and phytoplankton density tend to increase towards the end of the culture period, thus the diurnal fluctuation of dissolved oxygen also gradually increases. At the beginning of the culture period, nutrient levels and plant density
of phytoplankton are low, so dissolved oxygen levels are usually below saturation and fluctuate little. Towards the end of the culture period, phytoplankton growth causes significant fluctuations in dissolved oxygen; when phytoplankton grow excessively, the lowest dissolved oxygen level (early morning) will be below the fish's requirements, necessitating remedial measures.
Oxygen is the most important gas among the dissolved gases in the aquatic environment. It is essential for the life of organisms, especially aquatic organisms, because the diffusion coefficient of oxygen in water is much smaller than in air. According to Krogh (1919) (cited by Boyd, 1990), the diffusion coefficient of oxygen in air is 11, while in water it is only 34 x 10-6. Therefore, it easily leads to localized oxygen deficiency in the water body. Furthermore, in the hydrosphere, dissolved oxygen only accounts for 3.4% of the volume, while in the atmosphere, it accounts for up to 20.98% of the volume.
Therefore, the ideal dissolved oxygen concentration in water for shrimp and fish is above 5 ppm. However, if dissolved oxygen levels exceed saturation, fish will suffer from gas bubble disease in the blood and swim bladder, causing rupture or blockage of blood vessels leading to the brain, heart, and tissues throughout the fish's body, leading to the distension of the swim bladder and blood vessels, causing hemorrhage in the fins and anus. This analysis also confirms that this is also a major causative agent of the disease.
Source: Internet compilation Author: Minh Duc






